Pharmaceutical&Biotech
Industry
Understanding of the Industry
| The Features of Pharmaceutical Industry |
|---|
General pharmaceutical production is manufactured based on the semi-product and packaged by product unit |
Production type is divided by in-house production and outsourcing production (A consignment method is used for this case)
|
Synchronization of Quality and Production
|
Ethical-the-counter drugs are managed in the pharmaceutical serialization system
|
| The Future Paradigm of Pharmaceutical Industry |
|---|
There is a similar business content in the pharmaceutical, bio-tech, cosmetics, food industry
|
Structural Growth Potenntial – R&D
|
Establishment of Global Production System
|
Pharmaceutical/biotech industry issues & solutions
Sales Activity Management
- Marketing information about customers’ (potential/actual customers) marketing info, sales activity schedule, and activity history management

Handle Tasks on Mobile/Web
- When registering orders, it proceeds an automatic confirmation following the conditions or a separate authorization process through application of automatic check-logic

Link Customer Evaluation Info
- Bond risk management by connecting closed business info provided by NTS/Nice Information Service

Prescription Result Management
- A medical equipment supply agency enters prescription result data to proceed sales result analysis.

Maintain pharmaceutical compliance through expense report for economic gains from product briefing webinar and conferences.

Product Traceability Management
- It tracks Lot of entire process from production history → raw material input → purchase GR based on the GI Lot No

Health Insurance Review & Assessment Service Report
- Transmission of GI serial No. and supply details by linking ERP and pharmaceutical serialization system

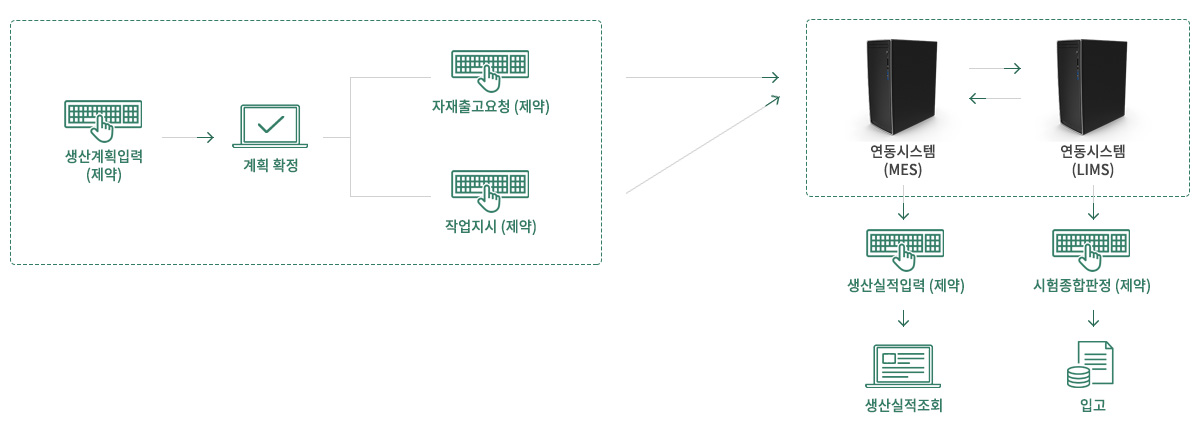
Link with Other System
- Transmission of production production/quality result through MES/LIMS link after manufacturing and package planning